A new type of banana shape bifunctional monomer of ester chalcones
DOI:
https://doi.org/10.11113/mjfas.v10n2.55Keywords:
Chalcones, banana shape liquid crystals, alkyl chains, esterAbstract
A synthesis of a new type of chalcone analogue banana-shaped liquid crystals compounds is described. The approach involved the reaction of hydroxyl chalcones with isophthaloyl dichloride to form benzene-1,3-dicarbonyl-bis-1-(4-alkoxyphenyl)-3-(4-hydroxylphenyl)prop-2-en-1-one (7a-c and 8a-c) having two chalcone units via esterification reaction. The hydroxyl chalcones, which differ in the length of alkyl group, CnH2n+1, where n= 10, 12 and 14 were synthesised via Claisen Schmidt condensation prior to the reaction with isophthaloyl dichloride. The reaction was a catalyst dependence. No sign of product was formed by employing NaH (60%). It was only occurred in the presence of NaH (95%) to afford banana shape bi-functional monomer of ester chalcone 7a-c and 8a-c. The synthesized compound was characterized by 1H and 13C Nuclear Magnetic Resonance (NMR), and Fourier Transform Infrared (FTIR).
________________________________________
GRAPHICAL ABSTRACT
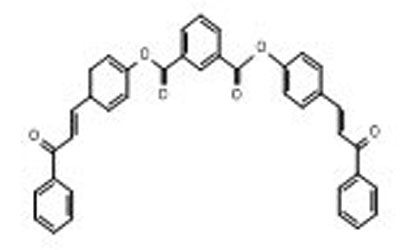
References
A. I. Jakli, (2008). United States Patent 20080002121.
D. H. Choi, Y. K. Cha, Bull. Korean Chem. Soc. 23 (2002) 587.
S. W. Nam, S. K. Kang, J. Y. Chang, Macromol. Res. 15 (2007) 74.
J. Indira, P. P. Karat, B. K. Sarojini, J. Cryst. Growth 242 (2002) 209.
B. T. Thaker, D. M. Patel, P. K. Tandel, M. S. Jesani, C. J. Vyas, A. D. Vansadia, Phase Transitions 78 (2005) 521.
K. H., Jung, S. Y. Hyun, D. M. Song, D. M. Shin, Opt. Mater. 21 (2002) 663.
B. Zhao, W. Q. Lu, Z. H. Zhou, Y. Wu, J. Mater. Chem. 10 (2000) 1513.
D. Fichou, T. Watanabe, T. Takeda, S. Miyata, Y. Goto, M. Nakayama, Jpn. J. Appl. Phys. 27 (1988) 429.
Y. Shimbo, Y. Takanishi, K. Ishikawa, E. Gorecka, D. Pociecha, J. Mieczkowski, K. Gomola, H. Takezoe, Jpn. J. Appl. Phys. 45 (2006) 282.
J. C. Rouillon, J. P. Marcerou, M. Laguerre, H. T. Nguyen, M. F. Achard, J. Mater. Chem. 11 (2001) 2946.
Z. Ngaini, N. I. Abdul Rahman, Can. J. Chem. 88 (2010) 654.
Z. Ngaini, N. I. Abdul Rahman, Phosphorus, Sulfur Silicon Relat. Elem. 185 (2010) 628.
B.A., Bhat, K. L. Dhar, S. C. Puri, A.K. Saxena, M. Shanmugavel, G.N. Qazi, Chem. Lett. 15 (2005) 3177.
K. Yoshizawa, and T. Shioiri, Tetrahedron Lett. 47 (2006) 4943.



















